 
Although LLZO satisfies the Monroe and Newman criteria and has exhibited promising properties, the failure of batteries that contain this electrolyte has been reported. One interesting electrolyte material for this application is Li 7La 3Zr 2O 12 (LLZO) because it has (a) a high voltage stability (up to 5 V), (b) high conductivity at room temperature (> 1 × 10 −4 S/cm), (c) low kinetic reactivity with Li, and (d) high shear modulus (approximately 55 GPa) 8, 9, 10, 11, 12. The failure and short circuit of batteries that contain ceramic electrolytes, however, have been reported 4, 5, 6, 7. Recently, the use of ceramics as solid electrolytes in all-solid Li metal batteries have attracted interest because of their high shear modulus. Monroe and Newman 3 have reported that a shear modulus that is twice that of Li can suppress dendrite growth. 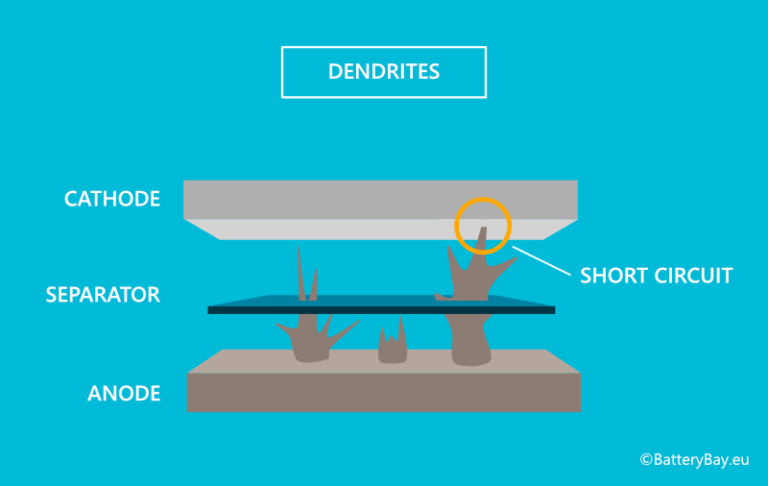
To overcome this limitation, one technique is the use of solid electrolytes with a high shear modulus to withstand perforations by dendrites and thus prevent battery short circuit. Lithium anodes, however, undergo dendrite formation during cycling that can increase the risk of battery short circuit 2. Metallic lithium is a potential anode material for high energy density Li-ion batteries because of its high capacity (3860 mAh g −1 for the reduced form) 1. Although the superior mechanical properties of LLZO make it an excellent electrolyte candidate for batteries, the further improvement of the electrochemical stabilization of the garnet–lithium metal interface is suggested. This work demonstrates the morphological and chemical evolution that occurs during cycling in a symmetrical Li–Li cell that contains LLZO. Moreover, the cross-section mapping comparison of the LLZO shows the inhomogeneous distribution of La, Zr, and C after cycling that was caused by lithium loss near the Li electrode and possible side reactions. The energy dispersive spectroscopy analyses of dendrites indicate the presence of Li, C, and O elements. Using the obtained SEM images, videos were created that show the inhomogeneous dissolution and deposition of lithium, which induce dendrite growth. In this work, in situ scanning electron microscopy (SEM) technique was employed to monitor the interface behavior between lithium metal and LLZO electrolyte during cycling with pressure. One promising candidate for this application is Li 7La 3Zr 2O 12 (LLZO) because it has excellent mechanical properties and chemical stability. In order to suppress dendrite growth, the use of electrolytes with a high shear modulus is suggested as an ionic conductive separator in batteries. This review may shed light on the critical understanding of current collector engineering for high-energy-density Li metal batteries.Dendrite formation, which could cause a battery short circuit, occurs in batteries that contain lithium metal anodes. Finally, the critical issues where there is incomplete understanding and the future research directions of Cu current collectors in practical LMAs are also prospected. Particularly, the design principles and strategies of functionalized Cu current collectors associated with their functionalities in optimizing Li plating/stripping behaviors are discussed. In this review, a comprehensive overview of Cu-based current collectors, including planar modified Cu foil, 3D architectured Cu foil and nanostructured 3D Cu substrates, for Li metal batteries is provided. 
Functionalized Cu current collectors have so far exhibited significant regulatory effects on stabilizing Li metal anodes (LMAs), and hold a great practical potential owing to their easy fabrication, low-cost and good compatibility with the existing battery technology. Li metal has been recognized as the most promising anode materials for next-generation high-energy-density batteries, however, the inherent issues of dendrite growth and huge volume fluctuations upon Li plating/stripping normally result in fast capacity fading and safety concerns. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
